In Epi-week 39, the Nigeria Centre for Disease Control (NCDC) was formally notified of suspected cases of monkey-pox disease in Bayelsa state. A response team was constituted immediately and deployed to the state to investigate the situation and verify the report. Preliminary investigative report revealed that some suspected cases of monkey-pox disease had been identified and possible routes of transmission as well. In collaboration with the Bayelsa State Government and other partners, the NCDC coordinated the initial response activities-isolation of patients, contact tracing, enhanced surveillance activities and commencement of public sensitization activities. Samples for laboratory testing were collected and results are being awaited.
There has been an increased awareness and sensitisation among Nigerians since the first news of suspected monkey-pox disease broke out in Nigeria. This has led to the identification of many suspected cases of the disease in seven additional states. This week’s editorial will digress from the components of yellow fever outbreak control (intended topic was to be on reactive vaccination) and focus on identifying and diagnosing a suspected case of monkey-pox. Some existing facts about the disease:
1. Monkey pox is a rare zoonotic disease caused by the monkey pox virus. The monkey pox virus belongs to the Orthopoxvirus genus in the family Poxviridae (Same family as smallpox and cowpox)
2. The disease derived its name after discovery in 1958 following identification in colonies of monkeys kept for research.
3. The natural reservoir of the disease remains unknown but rodents and monkeys are suspected to play a role in its transmission
4. Transmission of the disease occurs when a person is in contact with the virus from an animal, human or materials contaminated with the virus
5. Case fatality in monkey pox outbreaks has been between 1% and 10%, with most deaths occurring in younger age groups
6. There is no treatment or vaccine available although supportive treatment can be provided
Monkey pox disease has an incubation period of 6 to 16 days but can range from 5 to 21 days. Infection with the virus is divided into 2 periods:
1. Invasion Period
o Characterised by fever, intense headache, swelling of the lymph nodes (lymphadenopathy), back pain, muscle ache and intense lack of energy (asthenia)
2. Skin Eruption Period
o Various stages of the rash appears which begins on the face and spreads to other parts of the body. The face is affected in about 85% of cases and palms and soles are affected in 75% of cases.
o Rash evolves from maculopapular to vesicles, pustules and then crusts and this usually occurs within 10 days
Diagnosis of Monkey pox disease is through laboratory testing where the virus can be identified through a number of different tests such as enzyme-linked immunosorbent assay (ELISA) antigen detection test, polymerase chain reaction (PCR) assay and virus isolation by cell culture.
Prevention of the disease is through observing standard infection prevention and control measures in health care setting and hand hygiene measures in schools, homes, markets, and other public places.
The NCDC has since the onset of the suspected outbreak activated an Incident Management Structure in its Incident Coordination Centre (ICC) for coordination of response to the outbreak. This is in collaboration with partnering agencies and other relevant stakeholders. While awaiting the results of the laboratory samples, the NCDC enjoins all States to increase public awareness on the disease by dissemination of accurate information through authentic platforms. Members of the public are advised to seek information and get clarifications on myths about monkey pox disease from recognized health authorities in their communities and States. More information on Monkeypox can be found on http://ncdc.gov.ng/news/106/05-october-2017-%7C-monkeypox-advisory
1. cdc.gov/poxvirus/monkeypox
2. www.who.int
In the reporting week ending on the 1st of October, 2017:
o There were 218 new cases of Acute Flaccid Paralysis (AFP) reported. None was confirmed as Polio. The last reported case of Polio in Nigeria was in August 2016. Active case search for AFP is being intensified as Nigeria has assiduously reinvigorated its efforts at eradicating Polio.
o 20 suspected cases of Cholera were reported from seven LGAs (five States; Bauchi – 6, Borno – 1, Delta – 3, Kaduna – 5 and Kano – 5). No was laboratory confirmed and no death was recorded.
o One suspected case of Lassa fever was reported from Eti-0sa LGA in Lagos State. One was laboratory confirmed and no death was recorded.
o There were five suspected cases of Cerebrospinal Meningitis (CSM) reported from five LGAs in four States (Benue – 1, Enugu – 1, Gombe – 1 and Katsina - 2). Of these, none was laboratory confirmed and no death was recorded. Ongoing surveillance for CSM has been intensified in all the 26 States in the Nigeria meningitis belt.
o There were 244 suspected cases of Measles reported from 28 States. None was laboratory confirmed and no death was recorded.
In the reporting week, all States sent in their report. This is a remarkable improvement! Timeliness of reporting remains 84% in both previous and current weeks (Week 38 and 39) while completeness remains at 100%. It is very important for all States to ensure timely and complete reporting at all times, especially during an outbreak.
LASSA
Please note that the data reflects the routine reports i.e. all suspected cases including the laboratory positive and negative cases
1.1. One suspected case of Lassa fever with one Lab. Confirmed was reported from Eti-Osa LGA (Lagos State) in week 39, 2017 compared with zero case reported at the same period in 2016.
1.2. Laboratory results of the one suspected case was positive for Lassa fever and other VHFs (Lagos – 1).
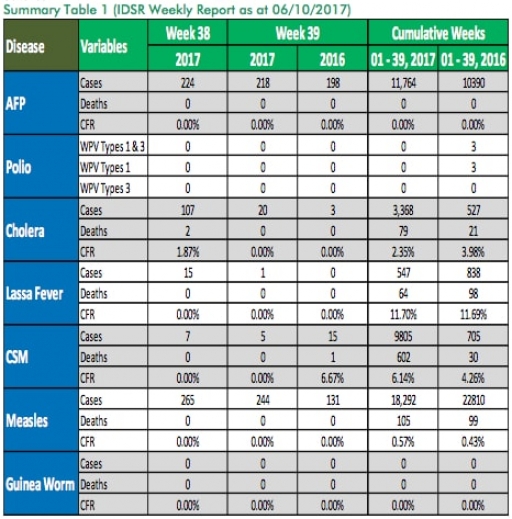
1.3. Between weeks 1 and 39 (2017), 547 suspected Lassa fever cases with 118 laboratory confirmed cases and 64 deaths (CFR, 11.70%) from 81 LGAs (26 States) were reported compared with 838 suspected cases with 83 laboratory confirmed cases and 98 deaths (CFR, 11.69%) from 135 LGAs (28 States) during the same period in 2016 (Figure 1).
1.4. Between weeks 1 and 52 2016, 921 suspected Lassa fever cases with 109 laboratory confirmed cases and 119 deaths (CFR, 12.92%) from 144 LGAs (28 States and FCT) were reported compared with 430 suspected cases with 25 laboratory confirmed cases and 40 deaths (CFR, 9.30%) from 37 LGAs (14 States and FCT) during the same period in 2015 (Figure 2).
1.5. Investigation and active case search ongoing in affected States with coordination of response activities by the NCDC with support from partners.
1.5.1. National Lassa Fever Working Group meeting and weekly National Surveillance and Outbreak Response meeting on-going at NCDC to keep abreast of the current Lassa fever situation in the country.
1.5.2. Response materials for VHFs provided to support States
1.5.3. New VHF guidelines have been developed by the NCDC (National Viral Haemorrhagic Fevers Preparedness guidelines, Infection Prevention and Control of VHF and Standard Operating Procedures for Lassa fever management) and are available on the NCDC website- http://ncdc.gov.ng/diseases/guidelines
1.5.4. VHF case-based forms completed by affected States are being entered into the new VHF management system. This system allows for the creation of a VHF database for the country.
1.5.5. Confirmed cases are being treated at identified treatment/isolation centres across the States with Ribavirin and necessary supportive management also instituted
1.5.6. Onsite support was earlier provided to Ogun, Nasarawa, Taraba, Ondo and Borno States by the NCDC and partners
1.5.7. Offsite support provided by NCDC/partners in all affected States
1.5.8. States are enjoined to intensify surveillance and promote Infection, Prevention and Control (IPC) measures in health facilities.
MEASLES
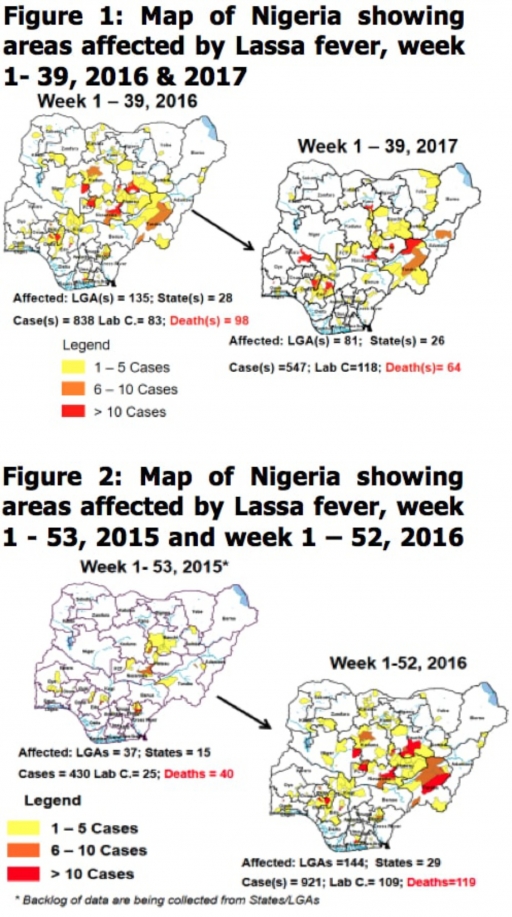
2.1. In the reporting week, 244 suspected cases of Measles were reported from 28 States compared with 131 suspected cases reported from 20 States during the same period in 2016.
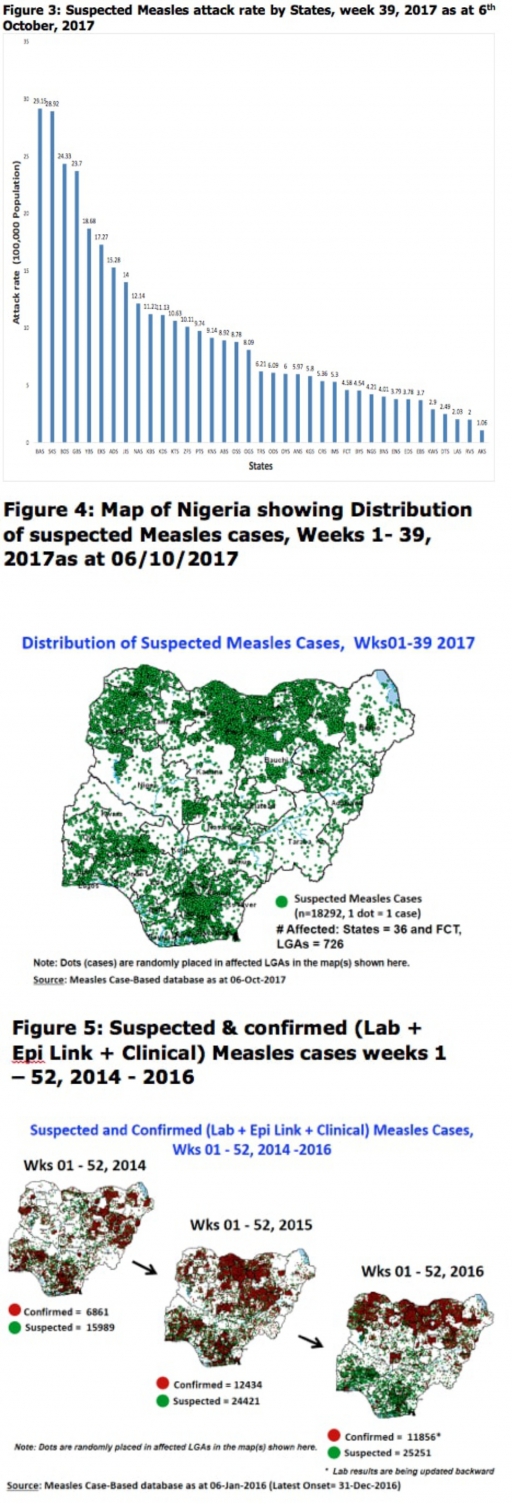
2.2. So far, 18,292 suspected Measles cases with 108 laboratory confirmed cases and 105 deaths (CFR, 0. 57%) have been reported in 2017 from 36 States and FCT (Figure 4) compared with 22,810 suspected cases and 99 deaths (CFR, 0.43%) from 36 States and FCT during the same period in 2016.
2.3. In 2016 (week 1 -52), 25,251 suspected Measles cases with 102 deaths (CFR, 0.40%) were reported from 36 States and FCT compared with 24,421 suspected cases with 127 deaths (CFR, 0.52%) during the same period in 2015 (Figure 5)
2.4. Response measures include immunisation for all vaccine-preventable diseases in some selected/affected wards/LGAs during SIAs, as well as case management.
2.5. Scheduled Measles campaigns in the North East were conducted from 12th – 17th January, 2017 in Adamawa, Borno and Yobe States (Phase I) and Phase II from 21st – 25th January, 2017 in Borno State and 4th – 8th February, 2017 in Yobe State
2.6. Measles Surveillance Evaluation and Establishment of the burden of Congenital Rubella Syndrome (CRS) in 12 selected States in the six geopolitical zones from the 17th -21st July 2017 conducted
2.6.1 Debrief meeting to review results and next steps from Measles evaluation conducted, held on the 15th of September 2017
2.7. Harmonisation of measles surveillance data with laboratory confirmed cases
POLIOMYELITIS
3.1. As at September 29th 2017, no new case of WPV was recorded
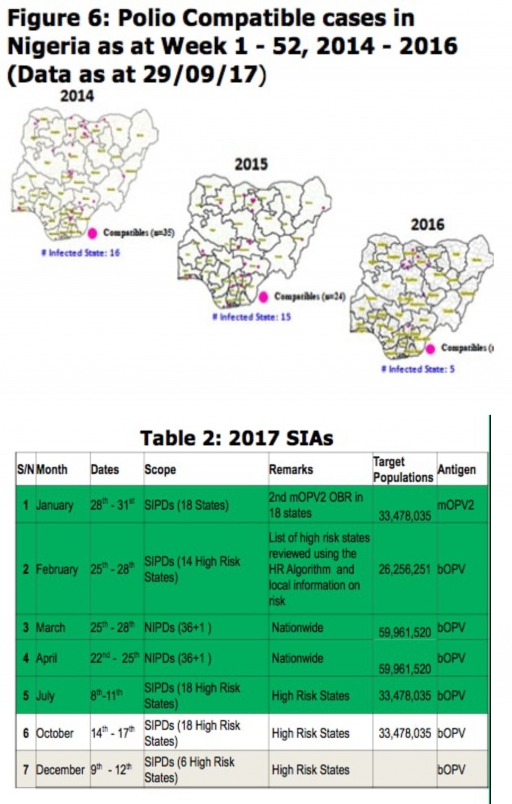
3.2. Three new cVDPV2, environmental derived and Polio compatible cases identified
3.2.1. In the reporting week, 218 cases of AFP were reported from 160 LGAs in 31 States and FCT
3.2.2. AFP Surveillance has been enhanced and outbreak response is on-going in Borno and other high risk States
3.2.3. The 1st round of SIPDs in 2017 was conducted from 28th – 31st January 2017 in the 18 high risk States. This was carried out using mOPV2 (2nd mOPV2 OBR). The schedule for other SIAs is as described in Table 2
3.2.4. The 2nd and 3rd round of SIPDs completed (25th-28th February and 8th – 11th July, 2017) in 14 & 18 high risk States using bOPV respectively.
3.2.5. The 1st and 2nd rounds of NIPDs completed (from 25th – 28th March, 2017 and 22nd – 25th April, 2017) nationwide respectively.
3.2.6. Between weeks 1 and 52 in 2016, four WPVs were isolated from Borno State compared to no WPV isolated during the same period in 2015.
3.3. No circulating Vaccine Derived Polio Virus type 2 (cVDPV2) was isolated in week 1 - 52, in both 2016 and 2015.
3.4. Between weeks 1 and 52, 2016 two (2) cVDPV2 were isolated in two LGAs (two States) while one (1) cVDPV2 was isolated from Kwali, FCT during the same period in 2015.
3.5. Six confirmed WPVs were isolated in 2014.
3.6. The SIAs were strengthened with the following events:
3.6.1. Immunisation for all vaccine-preventable diseases in some selected wards/LGAs.
3.6.2. Use of health camp facilities.
3.6.3. Field supportive supervision and monitoring.
3.6.4. Improved Enhanced Independent Monitoring (EIM) and Lots Quality Assessments (LQAs) in all Polio high risk States.
3.6.5. High level of accountability framework
CHOLERA
4.1. 20 suspected cases of Cholera were reported from seven LGAs (five States; Bauchi – 6, Borno – 1, Delta – 3, Kaduna - 5 and Kano – 5) in week 39 compared with three suspected cases reported from two LGAs in two States during the same period in 2016.
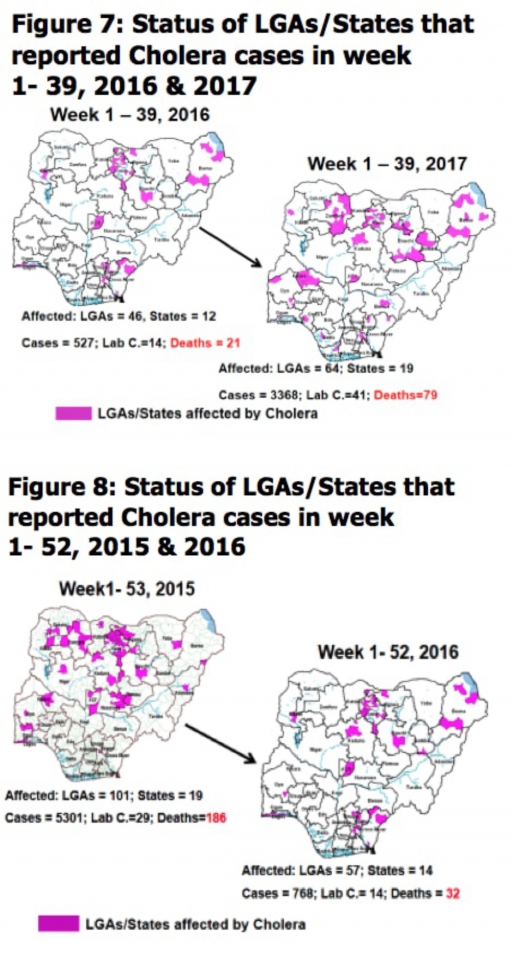
4.2. Between weeks 1 and 39 (2017), 3368 suspected Cholera cases with 41 laboratory confirmed and 79 deaths (CFR, 2.35%) from 64 LGAs (19 States) were reported compared with 527 suspected cases and 21 deaths (CFR, 3.98%) from 46 LGAs (12 States) during the same period in 2016 (Figure 7).
4.3. Between weeks 1 and 52 (2016), 768 suspected Cholera cases with 14 laboratory confirmed cases and 32 deaths (CFR, 4.17%) from 57 LGAs (14 States) were reported compared with 5,301 cases with 29 laboratory confirmed cases and 186 deaths (CFR, 3.51%) from 101 LGAs (18 States and FCT) during the same period in 2015 (Figure 8).
4.4. Cholera preparedness workshop held from 31st May – 1st June, 2017 in Abuja to
develop Cholera preparedness plan as the season set in.
4.5. NCDC/partners provided onsite support in Kwara, Zamfara and Kebbi States.
4.6 NCDC/partners are providing onsite support in Borno State.
4.7. Preparedness and Response to Acute Watery Diarrhoea/ Cholera Guidelines have been finalized: http://ncdc.gov.ng/themes/common/docs/protocols/45_1507196550.pdf
4.8. RDT procured by NCDC and WHO prepositioned in affected States
4.8. States are enjoined to intensify surveillance, implement WASH activities and ensure early reporting.
CEREBROSPINAL MENINGITIS (CSM)
5.7. In the reporting week 39, five suspected Cerebrospinal Meningitis (CSM) cases were reported from five LGAs (four States) compared with 15 suspected cases and one death (CFR, 6.67%) from five LGAs (four States) at the same period in 2016.
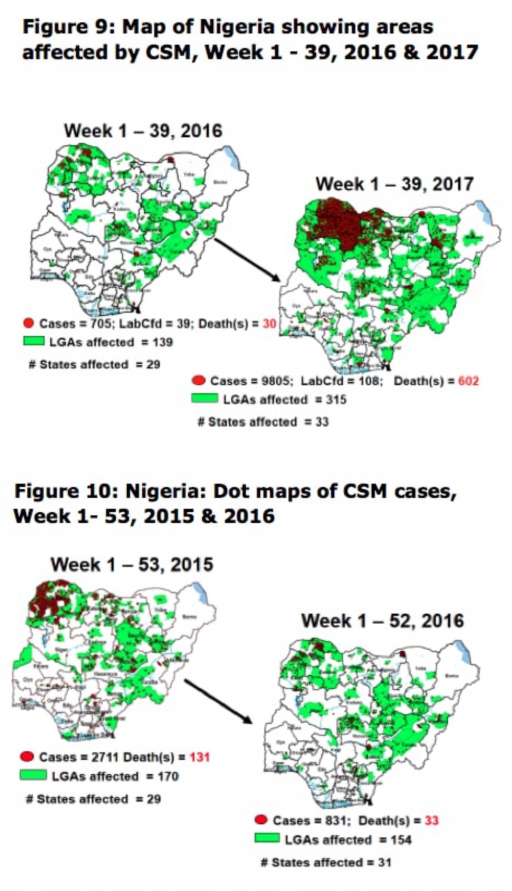
5.8. Between weeks 1 and 39 (2017), 9805 suspected CSM cases with 108 laboratory confirmed cases and 602 deaths (CFR, 6.14%) were recorded from 315 LGAs (33 States) compared with 705 suspected cases and 30 deaths (CFR, 4.26%) from 139 LGAs (29 States) during the same period in 2016 (Figure 9).
5.9. Between weeks 1 and 52, 2016, 831 suspected CSM cases with 43 laboratory confirmed cases and 33 deaths (CFR, 3.97%) were recorded from 154 LGAs (30 States and FCT) compared with 2,711 suspected cases and 131 deaths (CFR, 4.83%) from 170 LGAs (28 States and FCT) during the same period in 2015 (Figure 10)
5.10. Timeliness/completeness of CSM case-reporting from States to the National Level (2017 versus 2016): on average, 82.1% of the 26 endemic States sent CSM reports in a timely manner while 98.8% were complete in week 1 – 39, 2017 as against 86.0% timeliness and 99.7% completeness recorded within the same period in 2016
5.11. NCDC attended the 14th Annual Meeting on Surveillance, Preparedness and Response to Meningitis Outbreaks in Africa, and 4th Annual MenAfriNet Partners’ meeting held in Ouagadougou, Burkina Faso in preparation of 2017/2018 meningitis season from 12th to 15th September, 2017.
5.12. Ongoing finalisation of the National CSM Guidelines
5.13. Enhanced surveillance to begin 1st of December 2017, ahead of the 2017/2018 dry season
GUINEA WORM DISEASE
6.7. In the reporting week, no rumour report of Guinea Worm disease was received from any State.
6.8. Nigeria has celebrated eight consecutive years of zero reporting of Guinea worm disease in the country. The Country has been officially certified free of Dracunculiasis transmission by the International Commission for the Certification of Dracunculiasis Eradication (ICCDE).
(For further information, contact Nigeria Guinea Worm Eradication Program / Neglected Tropical Diseases Division, Public Health Department/Federal Ministry of Health)

Update on national Influenza sentinel surveillance, Nigeria week 1 - 39, 2017
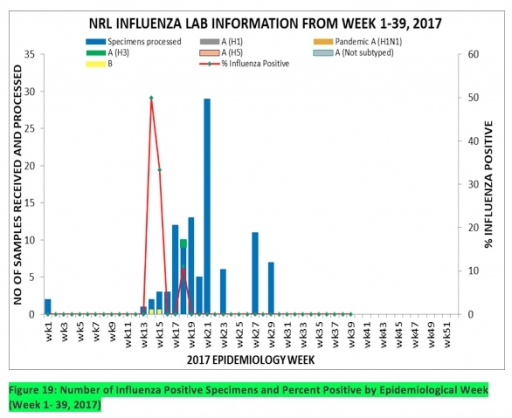
7.1. From week 1-39, a total of 103 suspected cases were reported, of which 95 were Influenza like-illness (ILI), 8 Severe Acute Respiratory Infection (SARI).
7.2 A total of 103 samples were received and all were processed. Of the processed samples, 95(92.2%) were ILI cases, 8(7.8%) were Severe Acute Respiratory Infection (SARI).
7.4. Of the 95 processed ILI samples, 1(1.05%) was positive for Influenza A; 2(2.1%) positive for Influenza B and 92(98.95%) were negative. Of the 8 processed SARI samples, none was positive for Influenza A and Influenza B.
7.5. In the reporting week 39, no samples were left unprocessed
FOR MORE INFORMATION CONTACT
Surveillance Unit:
Nigeria Centre for Disease Control,
801 Ebitu Ukiwe Street, Jabi, Abuja, Nigeria.
[email protected]
www.ncdc.gov.ng/reports
0800-970000-10










 Toll Free Number: 6232
Toll Free Number: 6232 Whatsapp: +234 708 711 0839
Whatsapp: +234 708 711 0839 SMS Number: +234 809 955 5577
SMS Number: +234 809 955 5577 