The current Cerebrospinal Meningitis outbreak in Nigeria has been ongoing for 17 weeks, since December 2016. The seasonal predilection of this outbreak helps to provide some room for preparedness and eventually, response. However, this current outbreak took on a different twist, as the causative strain identified is Neisseria meningitidis serotype C. In the past, Nigeria experienced CSM outbreaks mainly due to Neisseria meningitidis serotype A with this being common knowledge due to prompt and effective laboratory diagnosis of suspected cases.
Early detection of cases is important particularly if causative organisms may be attributable to different serotypes. In the current outbreak, suspected cases were identified early, but laboratory diagnoses were not carried out promptly, due to several reasons ranging from the absence of trained medical personnel for sample collection to the poor state of laboratories. Pre-knowledge of circulating strains of the organism would have played a critical role in mitigating the effect of the current outbreak.
In addition, laboratories are to ensure that results are sent out early to originating states so as to inform prompt institution of appropriate treatment for confirmed cases and commencement of response activities. A broad overview of this is to focus on early case identification and management
The Nigeria Centre for Disease Control (NCDC) continues to support all affected states in this outbreak and coordinate surveillance and response activities across the country. All States are enjoined to continue to ensure that laboratory confirmation of suspected cases of CSM, and indeed all epidemic-prone diseases is carried out promptly to inform response activities, in the event of an outbreak.
In the reporting week:
o There were 270 new cases of Acute Flaccid Paralysis (AFP) reported. None was confirmed as Polio. The last reported case of Polio in Nigeria was in August 2016. Active case search for AFP is being intensified as Nigeria has assiduously reinvigorated its efforts at eradicating Polio.
o No suspected case of Cholera was reported.
o There were 1517 suspected cases of Cerebrospinal Meningitis (CSM) reported from 113 LGAs in 23 States. Of these, 16 cases were laboratory confirmed and 114 deaths were recorded. Surveillance for CSM is ongoing and intensified in the States, particularly as the dry season has set in.
o There were 623 suspected cases of Measles reported from 31 States including the FCT. One death was recorded.
In the reporting week, three States (Adamawa, Edo and Oyo) failed to report while four States reported late. Timeliness of reporting increased from 77.0% in the previous week to 78.0% in the current week while completeness remains 99.0% in both previous and current weeks. It is very important for all States to ensure timely and complete reporting at all times.
1. Lassa fever
Please note that the data reflects the routine reports i.e. all suspected cases including the laboratory positive and negative cases
1.1. Four suspected cases of Lassa fever with one laboratory confirmed case and one death (CFR, 25.0%) were reported from three LGAs (three States) in week 14, 2017 compared with five suspected cases from two LGAs (Katsina State) during the same period in 2016.
1.2. Laboratory results of the 4 suspected cases were one positive for Lassa fever (Enugu – 1), one negative for Lassa fever and other VHFs (Kano - 1) while two results are pending (Ondo - 2).
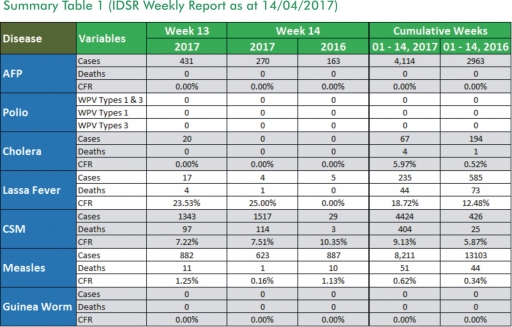
1.3. Between weeks 1 and 14 (2017), 235 suspected Lassa fever cases with 58 laboratory confirmed cases and 44 deaths (CFR, 18.72%) from 46 LGAs (17 States) were reported compared with 585 suspected cases with 58 laboratory confirmed cases and 73 deaths (CFR, 12.48%) from 115 LGAs (27 States) during the same period in 2016 (Figure 1).
1.4. Between weeks 1 and 52 2016, 921 suspected Lassa fever cases with 109 laboratory confirmed cases and 119 deaths (CFR, 12.92%) from 144 LGAs (28 States and FCT) were reported compared with 430 suspected cases with 25 laboratory confirmed cases and 40 deaths (CFR, 9.30%) from 37 LGAs (14 States and FCT) during the same period in 2015 (Figure 2).
1.5. Investigation and active case search ongoing in affected States with coordination of response activities by the NCDC with support from partners.
1.5.1. National Lassa Fever Working Group meeting and weekly National Surveillance and Outbreak Response meeting on-going at NCDC to keep abreast of the current Lassa fever situation in the country.
1.5.2. Response materials for VHFs prepositioned across the country by NCDC at the beginning of the dry season
1.5.3. New VHF guidelines have been developed by the NCDC (Interim National Viral Haemorrhagic fevers preparedness guidelines and Standard Operating Procedures for Lassa fever management)
1.5.4. Ongoing reclassification of reported Lassa fever cases
1.5.5. Ongoing review of the variables for case-based surveillance for VHF
1.5.6. VHF case-based forms completed by affected States are being entered into the new VHF management system. This system allows for the creation of a VHF database for the country.
1.5.7. Confirmed cases are being treated at identified treatment/isolation centers across the States with Ribavirin and necessary supportive management also instituted
1.5.8. Onsite support was earlier provided to Ogun, Nasarawa, Taraba and Ondo States by the NCDC and partners.
1.5.9. The NCDC deployed a team to Borno state to support the outbreak response and coordinate case management of patients and support implementation of IPC measures amongst healthcare workers
1.5.10. States are enjoined to intensify surveillance
2. MEASLES
2.1. In the reporting week, 623 suspected cases of Measles and 1 death (CFR, 0.16%) were reported from 30 States and FCT compared with 887 suspected measles cases and ten deaths (CFR, 1.13%) from 33 States and FCT during the same period in 2016.
2.2. So far, 8,211 suspected Measles cases with 55 laboratory confirmed cases and 51 deaths (CFR, 0. 62%) have been reported in 2017 from 36 states and FCT (Figure 4) compared with 13,103 suspected cases and 44 deaths (CFR, 0.34%) from 36 states and FCT during the same period in 2016.
2.3. In 2016 (week 1 -52), 25,251 suspected Measles cases with 102 deaths (CFR, 0.40%) were reported from 36 States and FCT compared with 24,421 suspected cases with 127 deaths (CFR, 0.52%) during the same period in 2015 (Figure 5)
2.4. Response measures include immunization for all vaccine-preventable diseases in some selected/affected wards/LGAs during SIAs, as well as case management.
2.5. Scheduled Measles campaign in the North East was conducted from 12th – 17th January, 2017 in Adamawa, Borno and Yobe States (Phase I) and Phase II from 21st – 25th January, 2017 in Borno State and 4th – 8th February, 2017 in Yobe State

3. POLIOMYELITIS
3.1. As at April 7th 2017, no new case of WPV recorded
3.2. Three new cVDPV2, environmental derived and Polio compatible cases identified
3.2.1. In the reporting week, 270 cases of AFP were reported from 198 LGAs in 31 States and FCT
3.2.2. AFP Surveillance has been enhanced and outbreak response is on-going in Borno and other high risk States
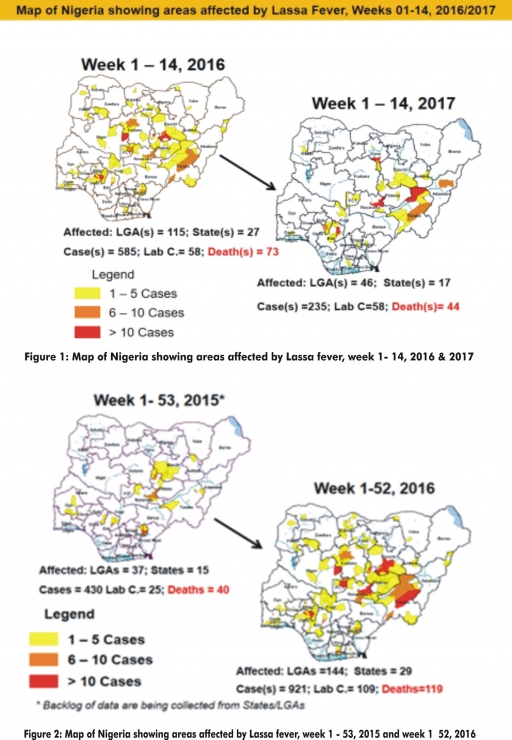
3.2.3. The 1st round of NIPDs in 2017 was conducted from 28th – 31st January 2017 in the 18 high risk States. This was carried out using mOPV2 (2nd mOPV2 OBR). The schedule for other SIAs is as described in Table 2
3.2.4. The 2nd round of SIPDs completed (25th-28th February, 2017) in 14 high risk States using bOPV.
3.2.5. The 3rd round of NIPDs completed (from 25th – 28th March, 2017) nationwide.
3.2.6. The 4th round of NIPDs is proposed for 22th – 25th April, 2017 nationwide.
3.2.7. Between weeks 1 and 52, 2016 four WPVs were isolated from Borno State compared to no WPV isolated during the same period in 2015.
3.3. No circulating Vaccine Derived Polio Virus type 2 (cVDPV2) was isolated in week 1 - 52, in both 2016 and 2015.
3.4. Between weeks 1 and 52, 2016 two (2) cVDPV2 were isolated in 2 LGAs (2 States) while one (1) cVDPV2 was isolated from Kwali, FCT during the same period in 2015.
3.5. Six confirmed WPVs were isolated in 2014.
3.6. The SIAs were strengthened with the following events:
3.6.1. Immunization for all vaccine-preventable diseases in some selected wards/LGAs.
3.6.2. Use of health camp facilities.
3.6.3. Field supportive supervision and monitoring.
3.6.4. Improved Enhanced Independent Monitoring (EIM) and Lots Quality Assessments (LQAs) in all Polio high risk States.
3.6.5. High level of accountability framework
4. CHOLERA
4.1. No suspected case of Cholera was reported in week 14 of both 2017 and 2016.
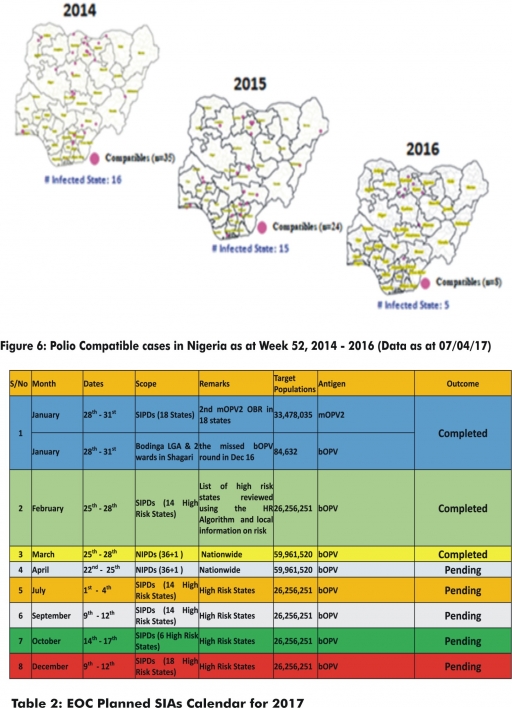
4.2. Between weeks 1 and 14 (2017), 67 suspected Cholera cases and four deaths (CFR, 5.97%) from ten LGAs (nine States) were reported compared with 194 suspected cases and 1 death (CFR, 0.52%) from 21 LGAs (seven States) during the same period in 2016 (Figure 7).
4.3. Between weeks 1 and 52 (2016), 768 suspected Cholera cases with 14 laboratory confirmed cases and 32 deaths (CFR, 4.17%) from 57 LGAs (15 States) were reported compared with 5,301 cases with 29 laboratory confirmed cases and 186 deaths (CFR, 3.51%) from 101 LGAs (18 States and FCT) during the same period in 2015 (Figure 8).
4.4. States are enjoined to intensify surveillance.
5. CEREBROSPINAL MENINGITIS (CSM)
5.1. In the reporting week, 1517 suspected Cerebrospinal Meningitis (CSM) cases with 16 laboratory confirmed cases and 114 deaths (CFR, 7.51%) were reported from 113 LGAs (23 States) compared with 29 suspected cases and three deaths (CFR, 10.35%) from 14 LGAs (nine States) during the same period in 2016.
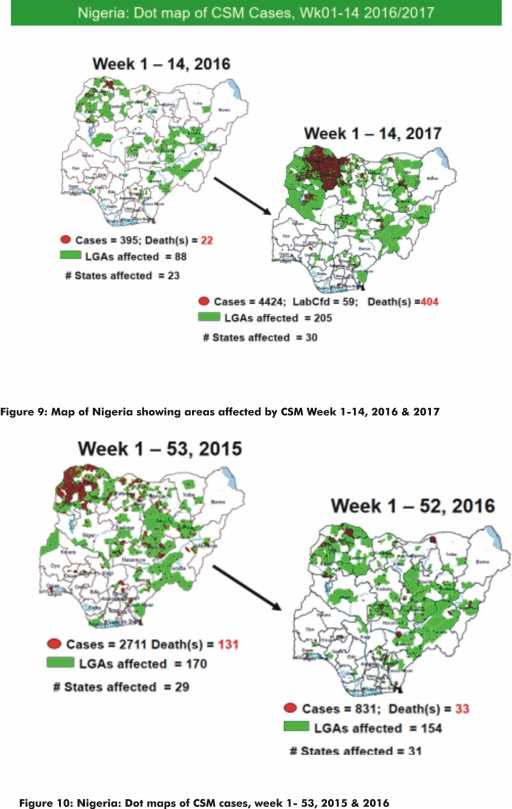
5.2. Between weeks 1 and 14 (2017), 4424 suspected CSM cases with 59 laboratory confirmed cases and 404 deaths (CFR, 9.13%) were recorded from 205 LGAs (30 States) compared with 426 suspected cases and 25 deaths (CFR, 5.87%) from 97 LGAs (24 States) during the same period in 2016 (Figure 9).
5.3. Between weeks 1 and 52, 2016, 831 suspected CSM cases with 43 laboratory confirmed cases and 33 deaths (CFR, 3.97%) were recorded from 154 LGAs (30 States and FCT) compared with 2,711 suspected cases and 131 deaths (CFR, 4.83%) from 170 LGAs (28 States and FCT) during the same period in 2015 (Figure 10)
5.4. Timeliness/completeness of CSM case-reporting from States to National Level (2017 versus 2016): on average, 78.0% of the 26 endemic States sent CSM reports in a timely manner while 94.8% were complete in week 1 – 14, 2017 as against 82.4% timeliness and 97.0% completeness recorded within the same period in 2016
5.5. CSM preparedness checklist sent to 36 States and FCT ahead of 2017 meningitis season completed
5.6. Confirmed cases are being treated at identified treatment centres in three States (Zamfara, Sokoto and Katsina) and necessary supportive management also instituted
5.7. Onsite support was earlier provided to Zamfara State and still ongoing.
5.8. Onsite support ongoing Sokoto, Katsina, Kebbi, Kano and Niger States by NCDC and partners
5.9. Intensive Surveillance is on-going in high risk States.
5.10. Reactive vaccination completed in Zamfara State for people aged one to 29 years.
5.11. Reactive vaccination ongoing in two wards (Gada and Kaffe) in Gada LGA in Sokoto State using polysaccharide meningococcal for aged two to 29 years.
5.12. Proposed reactive vaccination in Sokoto and Katsina States in progress using monosaccharide meningococcal conjugate.
6. GUINEA WORM DISEASE
6.1. In the reporting week, no rumour reports of Guinea Worm disease was received from any State.
6.2. Nigeria has celebrated eight consecutive years of zero reporting of Guinea worm disease in the country. The Country has been officially certified free of Dracunculiasis transmission by the International Commission for the Certification of Dracunculiasis Eradication (ICCDE).
(For further information, contact NIGEP NC/Director: Mrs. I, Anagbogu: +2348034085607, [email protected])
FOR MORE INFORMATION CONTACT
Surveillance Unit:
Nigeria Centre for Disease Control
801 Ebitu Ukiwe Street, Jabi, Abuja, Nigeria.
[email protected]
www.ncdc.gov.ng/reports
0800-970000-10









 Toll Free Number: 6232
Toll Free Number: 6232 Whatsapp: +234 708 711 0839
Whatsapp: +234 708 711 0839 SMS Number: +234 809 955 5577
SMS Number: +234 809 955 5577 