Hepatitis E is a liver disease caused by infection with a virus known as hepatitis E virus (HEV). Outbreaks of Hepatitis E usually occur as a result of faecal contamination of drinking water supplies. This tends to become more widespread during the rainy season.
As the rainy season continues in Nigeria with incidence of flood across some parts of the country, it is important for communities to be prepared for outbreaks of water-borne diseases such as Hepatitis E.
Like other outbreaks, response activities to an outbreak of Hepatitis E in States should be led by the State Ministry of Health, community oriented and carried out in a stepwise manner as seen below:
1. Prepare for an outbreak-This is important especially in emergency or high risk situations e.g. Refugee camps, Internally Displaced Persons camp
o Develop a basic plan or checklist for resource requirements in the event of an outbreak
o Set up a surveillance system to ensure an early warning once there is an increased incidence, particularly in vulnerable populations
o Develop a written plan for Hepatitis E covering roles and responsibilities, resources, skills and activities required
o Ensure standard treatment protocols are available to all health facilities and agencies
o Train clinical workers in case management, laboratory sample collection and transport, and water purification.
o Ensure stockpiles of essential treatment supplies are available such as intravenous fluids, laboratory sampling kits, transport media and water purification supplies.
o A competent laboratory is identified for confirmation of cases.
o Sources of additional supplies are identified.
2. Verify the diagnosis and confirm the existence of an outbreak.
3. Define a case and conduct case-finding.
4. Tabulate and orient data: time, place, person.
5. Take immediate control measures-categorized under three major activities
o Prevention of exposure: Controlling the source of infection to reduce the risk of the disease spreading to other members of the community. For hepatitis E outbreaks, this is the most important measure, and involves improving the quality and quantity of drinking water, treating and disposing of human waste correctly, improving personal hygiene and preparing safe and clean food.
o Prevention of infection: Focus should be placed on identifying pregnant women at health facilities, antenatal clinics and other points. Efforts should be targeted at reducing the risk among pregnant women since the infection is more likely to lead to severe disease and death in such women than in the general population. These women should be a priority group for provision of safe water and good sanitation.
o Prevention of death: Deaths can be minimized through prompt diagnosis and management of cases, including timely referral to a health-care facility. It may be important to avoid administration of unnecessary drugs, as these may be hepatotoxic; such drugs are likely to be particularly harmful for patients with acute hepatitis E.
6. Communicate findings.
7. Implement and evaluate control measures-entails instituting longer term control measures to end the current outbreak and prevent future outbreaks. These measures should be more extensive than earlier control measures and evaluated for effectiveness.
Examples of such measures are: recommending different food safety procedures in public eating places and implementing a better chlorination programme for public water systems.
After an outbreak, the outbreak control team must carry out a thorough evaluation of the response to the particular outbreak, including identification of its cause(s), surveillance and detection, level of preparedness, management and control measures during the outbreak. The specific issues that should be evaluated under each heading include the timeliness of detection and adequacy of response, effectiveness, cost, lost opportunities and new/revised policies. The findings of this evaluation should be documented in a written report that contains clear recommendations on the epidemiological characteristics of the epidemic, surveillance, preparedness and control measures carried out
The Nigeria Centre for Disease Control (NCDC) urges all States to commence early preparedness plans for a more coordinated response system in the event of a Hepatitis E outbreak. The NCDC will continue to work with other partners to provide support in outbreak coordination in any affected state.
States are advised to create increased awareness on Hepatitis E infection in their communities and implement preventive measures, in view of the heavy rainfalls recorded recently in the country.
1. Water Borne Outbreaks of Hepatitis E: Recognition, investigation and control. Technical Report. Geneva. WHO.2014
2. https://www.cdc.gov/hepatitis/statistics/surveillanceguidelines.htm
o There were 393 new cases of Acute Flaccid Paralysis (AFP) reported. None was confirmed as Polio. The last reported case of Polio in Nigeria was in August 2016. Active case search for AFP is being intensified as Nigeria has assiduously reinvigorated its efforts at eradicating Polio.
o 81 suspected cases of Cholera were reported from eight LGAs in five States with three laboratory confirmed cases and three deaths.
o There were 12 suspected cases of Cerebrospinal Meningitis (CSM) reported from ten LGAs in ten States. Of these, none was laboratory confirmed and no death was recorded. Ongoing surveillance for CSM has been intensified in the States.
o There were 281 suspected cases of Measles reported from 33 States. None was laboratory confirmed and no death was recorded.
In the reporting week ending on the 23rd of July, 2017:
o There were 393 new cases of Acute Flaccid Paralysis (AFP) reported. None was confirmed as Polio. The last reported case of Polio in Nigeria was in August 2016. Active case search for AFP is being intensified as Nigeria has assiduously reinvigorated its efforts at eradicating Polio.
o 81 suspected cases of Cholera were reported from eight LGAs in five States with three laboratory confirmed cases and three deaths.
o There were 12 suspected cases of Cerebrospinal Meningitis (CSM) reported from ten LGAs in ten States. Of these, none was laboratory confirmed and no death was recorded. Ongoing surveillance for CSM has been intensified in the States.
o There were 281 suspected cases of Measles reported from 33 States. None was laboratory confirmed and no death was recorded.
In the reporting week, Gombe State failed to send in any report. Timeliness of reporting remains at 82% in both previous and current weeks (Week 28 and 29) while completeness also remains at 100%. It is very important for all States to ensure timely and complete reporting at all times, especially during an outbreak.
1. Lassa fever
Please note that the data reflects the routine reports i.e. all suspected cases including the laboratory positive and negative cases
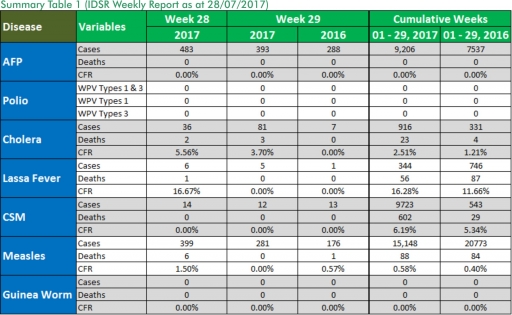
1.1. Five suspected cases of Lassa fever were reported from five LGAs (Plateau State) in week 29, 2017 compared with one case from Abuja Municipal Area Council (FCT) at the same period in 2016.
1.2. Laboratory results of the five suspected cases are four positives for Lassa fever while one was negative for Lassa fever and other VHF.
1.3. Between weeks 1 and 29 (2017), 344 suspected Lassa fever cases with 86 laboratory confirmed cases and 56 deaths (CFR, 16.28%) from 67 LGAs (22 States) were reported compared with 746 suspected cases with 72 laboratory confirmed cases and 87 deaths (CFR, 11.66%) from 127 LGAs (27 States) during the same period in 2016 (Figure 1).
1.4. Between weeks 1 and 52 2016, 921 suspected Lassa fever cases with 109 laboratory confirmed cases and 119 deaths (CFR, 12.92%) from 144 LGAs (28 States and FCT) were reported compared with 430 suspected cases with 25 laboratory confirmed cases and 40 deaths (CFR, 9.30%) from 37 LGAs (14 States and FCT) during the same period in 2015 (Figure 2).
1.5. Investigation and active case search ongoing in affected States with coordination of response activities by the NCDC with support from partners.
1.5.1. National Lassa Fever Working Group meeting and weekly National Surveillance and Outbreak Response meeting on-going at NCDC to keep abreast of the current Lassa fever situation in the country.
1.5.2. Response materials for VHFs prepositioned across the country by NCDC at the beginning of the dry season
1.5.3. New VHF guidelines have been developed by the NCDC (Interim National Viral Haemorrhagic Fevers Preparedness guidelines and Standard Operating Procedures for Lassa fever management) and are available on the NCDC website.
1.5.4. Ongoing reclassification of reported Lassa fever cases
1.5.5. Ongoing review of the variables for case-based surveillance for VHF
1.5.6. VHF case-based forms completed by affected States are being entered into the new VHF management system. This system allows for the creation of a VHF database for the country.
1.5.7. NCDC team sent to Edo State to support Lassa fever data harmonization & Updating of VHF case-based management database
1.5.8. Confirmed cases are being treated at identified treatment/isolation centres across the States with Ribavirin and necessary supportive management also instituted
1.5.9. Onsite support was earlier provided to Ogun, Nasarawa, Taraba, Ondo and Borno States by the NCDC and partners
1.5.10. Offsite support provided by NCDC/partners in all affected States
1.5.11. NCDC and partners are providing onsite support in Ondo and Plateau State
1.5.12. States are enjoined to intensify surveillance and promote Infection, Prevention and Control (IPC) measures in health facilities.
2. MEASLES
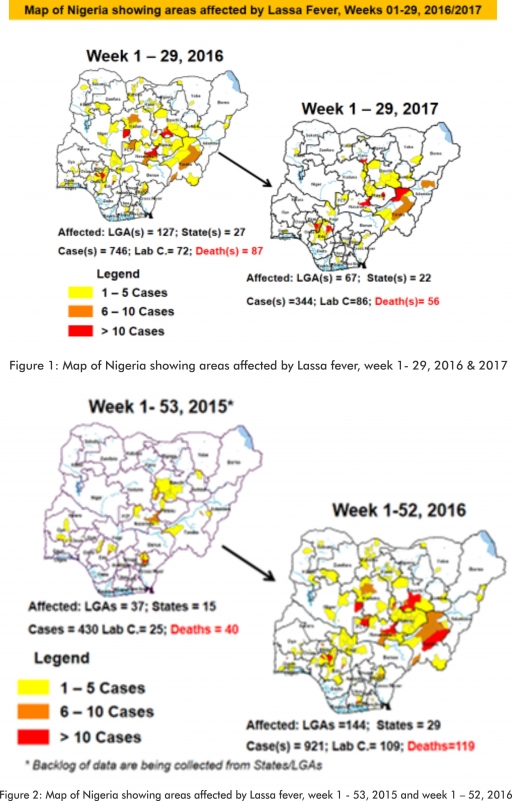
2.1. In the reporting week, 281 suspected cases of Measles were reported from 33 States compared with 176 suspected measles cases and one death (CFR, 0.57%) reported from 25 States during the same period in 2016.
2.2. So far, 15,148 suspected Measles cases with 108 laboratory confirmed cases and 88 deaths (CFR, 0. 59%) have been reported in 2017 from 36 States and FCT (Figure 4) compared with 20,773 suspected cases and 84 deaths (CFR, 0.40%) from 36 States and FCT during the same period in 2016.
2.3. In 2016 (week 1 -52), 25,251 suspected Measles cases with 102 deaths (CFR, 0.40%) were reported from 36 States and FCT compared with 24,421 suspected cases with 127 deaths (CFR, 0.52%) during the same period in 2015 (Figure 5)
2.4. Response measures include immunization for all vaccine-preventable diseases in some selected/affected wards/LGAs during SIAs, as well as case management.
2.5. Scheduled Measles campaigns in the North East were conducted from 12th – 17th January, 2017 in Adamawa, Borno and Yobe States (Phase I) and Phase II from 21st – 25th January, 2017 in Borno State and 4th – 8th February, 2017 in Yobe State
2.6. Measles Surveillance Evaluation and Establishment of the burden of Congenital Rubella Syndrome (CRS) in 12 selected States in the six geopolitical zones from the 17th -21st July 2017
2.7. Harmonization of measles surveillance data with laboratory confirmed cases
3. POLIOMYELITIS
3.1. As at July 16th 2017, no new case of WPV was recorded
3.2. Three new cVDPV2, environmental derived and Polio compatible cases identified
3.2.1. In the reporting week, 393 cases of AFP were reported from 273 LGAs in 34 States and FCT
3.2.2. AFP Surveillance has been enhanced and outbreak response is on-going in Borno and other high risk States
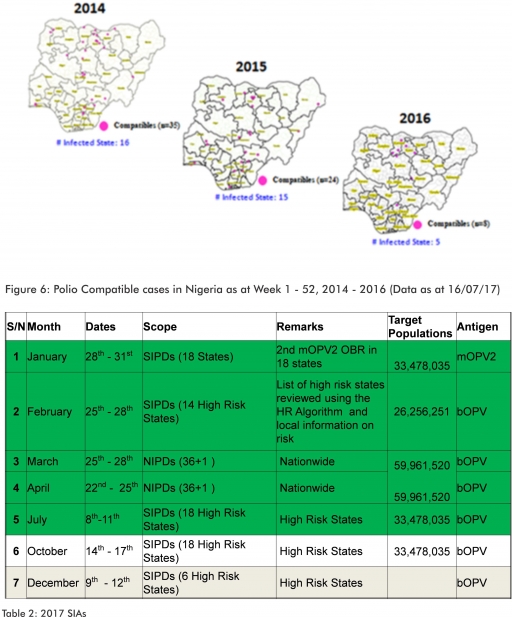
3.2.3. The 1st round of SIPDs in 2017 was conducted from 28th – 31st January 2017 in the 18 high risk States. This was carried out using mOPV2 (2nd mOPV2 OBR). The schedule for other SIAs is as described in Table 2
3.2.4. The 2nd and 3rd round of SIPDs completed (25th-28th February and 8th – 11th July, 2017) in 14 & 18 high risk States using bOPV respectively.
3.2.5. The 1st and 2nd rounds of NIPDs completed (from 25th – 28th March, 2017 and 22nd – 25th April, 2017) nationwide respectively.
3.2.6. Between weeks 1 and 52 in 2016, four WPVs were isolated from Borno State compared to no WPV isolated during the same period in 2015.
3.3. No circulating Vaccine Derived Polio Virus type 2 (cVDPV2) was isolated in week 1 - 52, in both 2016 and 2015.
3.4. Between weeks 1 and 52, 2016 two (2) cVDPV2 were isolated in two LGAs (two States) while one (1) cVDPV2 was isolated from Kwali, FCT during the same period in 2015.
3.5. Six confirmed WPVs were isolated in 2014.
3.6. The SIAs were strengthened with the following events:
3.6.1. Immunization for all vaccine-preventable diseases in some selected wards/LGAs.
3.6.2. Use of health camp facilities.
3.6.3. Field supportive supervision and monitoring.
3.6.4. Improved Enhanced Independent Monitoring (EIM) and Lots Quality Assessments (LQAs) in all Polio high risk States.
3.6.5. High level of accountability framework
4. CHOLERA
4.1. 81 suspected cases of Cholera cases with three laboratory confirmed and three deaths (CFR, 3.70%) were reported from eight LGAs (five States) in week 29 compared with seven suspected cases from Bichi LGA (Kano State) at the same period in 2016.
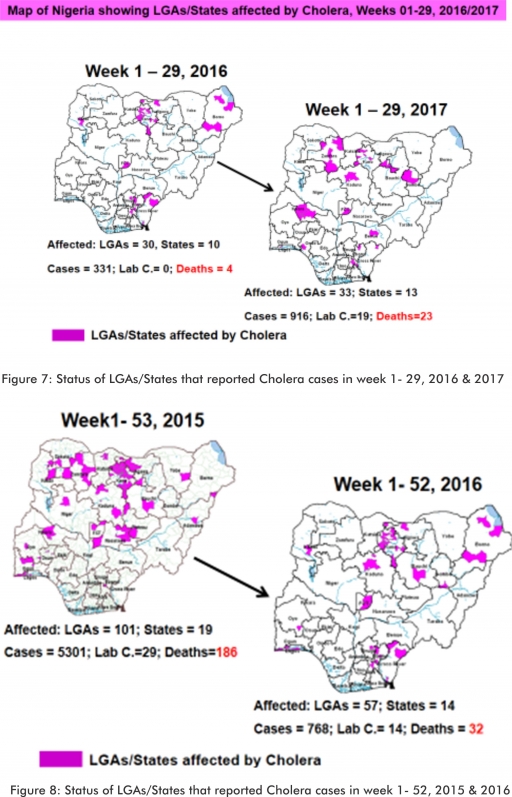
4.2. Between weeks 1 and 29 (2017), 916 suspected Cholera cases with 19 laboratory confirmed and 23 deaths (CFR, 2.51%) from 33 LGAs (13 States) were reported compared with 331 suspected cases and four deaths (CFR, 1.21%) from 30 LGAs (ten States) during the same period in 2016 (Figure 7).
4.3. Between weeks 1 and 52 (2016), 768 suspected Cholera cases with 14 laboratory confirmed cases and 32 deaths (CFR, 4.17%) from 57 LGAs (14 States) were reported compared with 5,301 cases with 29 laboratory confirmed cases and 186 deaths (CFR, 3.51%) from 101 LGAs (18 States and FCT) during the same period in 2015 (Figure 8).
4.4. Cholera preparedness workshop held from 31st May – 1st June, 2017 in Abuja to
develop Cholera preparedness plan as the season set in.
4.5. NCDC/partners provided onsite support in Kwara State.
4.6 NCDC/partners are providing onsite support in Zamfara State.
4.7 States are enjoined to intensify surveillance, implement WASH activities and ensure early reporting.
5. CEREBROSPINAL MENINGITIS (CSM)
5.1. In the reporting week 29, 12 suspected Cerebrospinal Meningitis (CSM) cases were reported from ten LGAs (ten States) compared with 13 suspected cases from nine LGAs (eight States) at the same period in 2016.
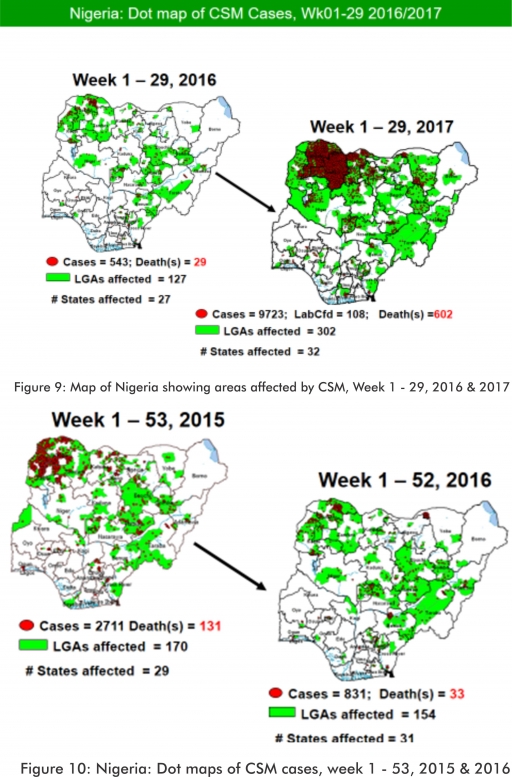
5.2. Between weeks 1 and 29 (2017), 9723 suspected CSM cases with 108 laboratory confirmed cases and 602 deaths (CFR, 6.19%) were recorded from 302 LGAs (32 States) compared with 543 suspected cases and 29 deaths (CFR, 5.34%) from 127 LGAs (27 States) during the same period in 2016 (Figure 9).
5.3. Between weeks 1 and 52, 2016, 831 suspected CSM cases with 43 laboratory confirmed cases and 33 deaths (CFR, 3.97%) were recorded from 154 LGAs (30 States and FCT) compared with 2,711 suspected cases and 131 deaths (CFR, 4.83%) from 170 LGAs (28 States and FCT) during the same period in 2015 (Figure 10)
5.4. Timeliness/completeness of CSM case-reporting from States to the National Level (2017 versus 2016): on average, 80.6% of the 26 endemic States sent CSM reports in a timely manner while 98.4% were complete in week 1 – 29, 2017 as against 84.1% timeliness and 99.2% completeness recorded within the same period in 2016
5.5. CSM preparedness checklist sent to 36 States and FCT ahead of 2017 meningitis season
5.6. Confirmed cases treated at identified treatment centres in affected States (Zamfara, Sokoto, Katsina, Kebbi, Niger, Kano, Yobe and Jigawa) and necessary supportive management also instituted
5.7. CSM National Emergency Operations Centre constituted at the Nigeria Centre for Disease Control
5.8. Onsite support provided to Zamfara, Sokoto, Katsina, Kebbi, Kano, Yobe and Niger States by NCDC and partners
5.9. Off-site support provided to other States
5.10. Intensive Surveillance in high risk States.
5.11. Reactive vaccination completed in Zamfara State for people aged one to 29 years using polysaccharide meningococcal A & C vaccine.
5.12. Reactive vaccination completed in two wards (Gada and Kaffe) in Gada LGA in Sokoto State using polysaccharide meningococcal A & C vaccine for people aged two to 29 years.
5.13. Reactive vaccination completed in nine LGAs in Sokoto State using monosaccharide meningococcal conjugate C vaccine for aged one to 20 years.
5.14. Reactive vaccination campaign completed in Yobe State for people aged two to 29 years using polyvalent ACW conjugate vaccine.
5.15. Medical teams were trained and deployed to support case management in Sokoto and Zamfara States completed (from Friday 5th - 26th May, 2017).
5.16. Deployed mobile testing laboratory to Zamfara State to aid diagnosis
5.17. A Team was deployed by NCDC/WHO to support surveillance activities, laboratory data harmonization and monitoring of the implementation plan in Yobe state
5.18. National CSM EOC has been stepped down
5.19. Evaluation of the CSM outbreak response in Zamfara and Sokoto States is ongoing by NCDC and WHO
5.20. National CSM After-Action Review meeting conducted in Sokoto State from the 24th – 25th of July 2017.
6. GUINEA WORM DISEASE
6.1. In the reporting week, no rumour report of Guinea Worm disease was received from any State.
6.2. Nigeria has celebrated eight consecutive years of zero reporting of Guinea worm disease in the country. The Country has been officially certified free of Dracunculiasis transmission by the International Commission for the Certification of Dracunculiasis Eradication (ICCDE).
(For further information, contact NIGEP NC/Director: Mrs. I, Anagbogu: +2348034085607, [email protected])
FOR MORE INFORMATION CONTACT
Surveillance Unit:
Nigeria Centre for Disease Control
801 Ebitu Ukiwe Street, Jabi, Abuja, Nigeria.
[email protected]
www.ncdc.gov.ng/reports
0800-970000-10










 Toll Free Number: 6232
Toll Free Number: 6232 Whatsapp: +234 708 711 0839
Whatsapp: +234 708 711 0839 SMS Number: +234 809 955 5577
SMS Number: +234 809 955 5577 